Key Insights:
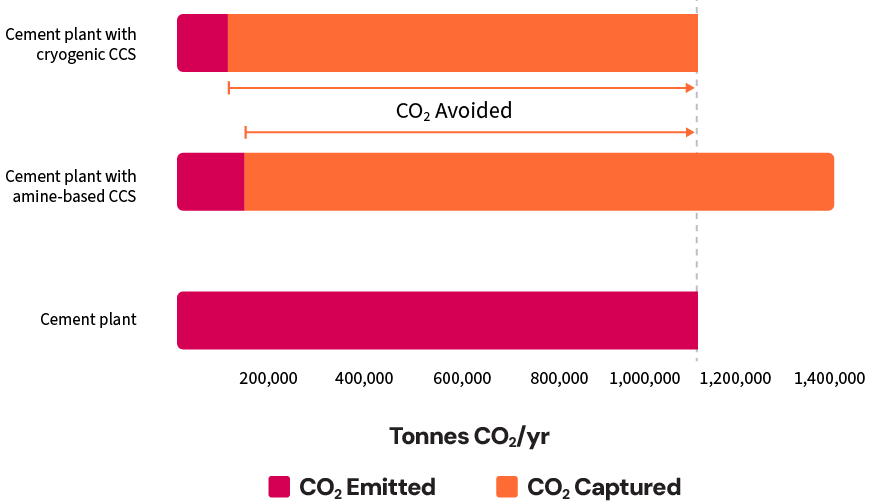
- CO2 Avoided (Emissions Reduction): Both technologies significantly reduced CO2 emissions, but the cryogenic scenario achieved a slightly higher overall emissions reduction (i.e., 90% compared to 87% for the amine-based system). This difference is due to the amine system’s use of an auxiliary boiler, which introduced additional CO2 emissions. Although the flue gas from the auxiliary boiler is sent to the capture plant, only 90% of those emissions were captured, leaving 10% uncaptured and contributing to the lower overall emissions reduction.
- CO2 Captured: The amine-based scenario captured approximately 30% more CO2 than the cryogenic scenario. This is because it also captured CO2 emissions from the auxiliary boiler used to generate steam for solvent regeneration, which is not required in the cryogenic scenario.